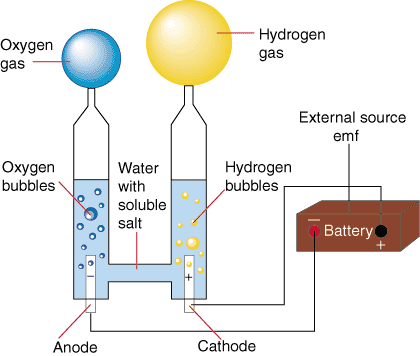
Explanation of Hydrogen Cells
hydrogen can not be broken down into any simpler forms. it is found naturally in the world, and theirs more than plenty to go around. the best thing about hydrogen is, that its is very powerful energy source that pollutes very little. A fuel cell combines hydrogen and oxygen to produce electricity, heat, and water. it is a great electrical power source for electric motors propelling vehicles. that's exactly what we did in our group. We used hydrogen cells along with solar panels to power our car. The way we did it is we filled two side of a hydrogen cells with water, and placed a cap on top with out any air bubbles. We then filled the entire cells and waited for ten minutes. After the ten minutes we connected the cells to a solar panel and placed the panel in front of a spotlight. the panels would gather the heat, and the cells would convert the heat into electricity. From there we used a grids and circuits to transfer the electricity from the cell to the car motor.
Explanation of solar Panels
solar panels are applications that convert light(sunlight) into usable electricity. solar panels are a collection of solar cells, and if you gather enough of them you can produce enough energy to power an application of your choice. the more light that hits the cells, the more energy it produces. the good thing about solar panels are that they give off zero pollution, although they are costly. Also they aren't the most efficient power source. Although in our group that what we used to power our car. When we hade the hydrogen cell ready, we connect that to the solar panels and we placed it in front of a light. The light was a replacement for the sun, since we were building indoors. The panel would collect the light energy and send it to the hydrogen cell. there the energy would be converted to electricity and sent to the car.
Typed Packet
1. Read the Fuel Cell User Guide.
2. Follow the directions in the Fuel Cell User Guide under the section Preparing the Fuel Cell for Use.
3. Shine a bright light source on the solar panel, always keeping at least 8 inches of separation between the two to avoid melting the solar module plastic.
Set your multimeter to measure voltage and connect the multimeter test leads to the solar panel terminals. Move the solar panel or light source to determine the location that produces the highest voltage value. You may want to mark the positions with some tape. Record the open-circuit voltage. Note the current is zero, since a voltmeter has nearly infinite resistance.
VOC = Open-Circuit Voltage ___1.51 volts ___Power = VOC x 0 A = 0 W
4. With the test leads disconnected, set your multimeter to measure current. Return the solar module to the same exact position that produced the highest voltage value and measure the current. Record this short-circuit current. Note that the voltage is zero, since an ammeter has nearly zero resistance.
ISC = Short-Circuit Current ____111 ma_____ Power = 0 V x ISC = 0 W
5. Calculate the amount of power that would be produced by the solar module if it could simultaneously produce the voltage and current you measured in the previous two steps.
For this illumination level, the solar module will deliver, at most, about 70% of this theoretical maximum, and will do so at a resistance between zero and infinite resistance.
Maximum Theoretical Power = VOC x ISC = ____167.610 w___
6. Attach the solar panel to the solar hydrogen automobile. Using a standoff or another suitable method, prop up one end of the chassis so that the motor-driven wheel is not in contact with the ground. Connect the motor leads to the solar module using the breadboard to make the connections. Position the light source to produce maximum voltage leaving a minimum distance of 8 inches between solar module and the lamp. Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? _____no____
7. Set your multimeter to measure voltage. Connect the multimeter test leads to the solar module terminals. Record the load voltage value.(Drive gear should be engaged)
V = Load Voltage __________
8. Disconnect the test leads and set your multimeter to measure current. Connect the multimeter in series with the solar module. Record the load current.
I = Load Current = __________
9. Calculate the power delivered by the solar module when it is loaded by the motor with the wheels off the ground.
P = Load Power = I V = __________ for solar module.
10. Energize the fuel cell by using one of the power sources according to the directions in the Fuel Cell User Guide under the section Powering the Fuel Cell (Electrolysis).
Fuel cells can be damaged by high current. If using a DC power supply with the Heliocentris fuel cell, do not use more than 500 mA. Do not use a battery to energize the fuel cell.
11. After the fuel cell is energized, attach the fuel cell to the motor using the breadboard to make the connections. Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? ______
12. With the test leads disconnected, set the multimeter to measure voltage. Connect the multimeter test leads to the fuel cell terminals. Record the voltage value.
V = Load Voltage __________
13. With the test leads disconnected, set the multimeter to measure 10 A current, using the 10 A meter receptacle. Connect the test leads in series with the fuel cell.
Caution! Never measure current from the fuel cell without a resistor, motor, or other load in series with the ammeter. Doing so can permanently damage the fuel cell.
Record the current value. Load Current = __________
14. Calculate the power delivered by the fuel cell. P = Load Power = I V = __________ for fuel cell.
15. Remove the fuel cell and solar module and attach the two AAA battery holders to your vehicle using zip ties. Using the breadboard, connect the batteries in series with each other and with the motor. (See next step for wiring hints.) Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? __yes____
16. With the test leads disconnected, set the multimeter to measure voltage. Connect the multimeter test leads to the motor terminals. Record the voltage value.
V = Load Voltage ____5.6 v______
17. With the test leads disconnected, set the multimeter to measure 10 A current, using the 10 A meter receptacle. Connect the test leads in series with the motor terminals. Record the current value.
Load Current = _____140 ma_____
18. Calculate the power delivered by the batteries in series. P = Load Power = I V = ____.784 w______ for batteries in series
19. Using the breadboard, connect the batteries in parallel with each other and with the motor. Is there enough power to turn the motor? If so, is there enough power to turn the motor with the wheels on the ground? __yes____
20. With the test leads disconnected, set the multimeter to measure voltage. Connect the multimeter test leads to the motor terminals. Record the voltage value.
V = Load Voltage _____.18 v_____
21. With the test leads disconnected, set the multimeter to measure 10 A current, using the 10 A meter receptacle. Connect the test leads in series with the motor terminals. Record the current value.
Load Current = _____25 ma_____
22. Calculate the power delivered by the batteries in parallel.
P = Load Power = I V = ____.0045 w______ for batteries in parallel
Explanation of Series and Parallel Circuits
A series circuit is a closed system were within that system the current can only travel in one path. even though that circuit can have more than one power supply, and more than one resistor inside the circuit. The current is forced to move in one direction, and the direction of that movement is from the negative side of the power supply to the positive side. The thing about the series circuit is if you have more than one applications on the circuit(for example 3 light bulbs), if you remove the first one the circuit is broken. Therefore the other two applications stop working too. On the other hand the parallel circuit has more than one path for the current to travel. Although it's still closely related to the series. Parallel also can have more than one power supply, and resistors on the circuit. Except this time when one of the applications in the circuit is removed or breaks. The other ones can still function because the current is flowing through different path. So in some cases the parallel works better, but doesn't always give the same amount of power as series.
Picture Of Circuit
Pictorial Representations of Circuit Connections
Reflection
Overall this was not that great of a project. Although i had a great time working with Anthony and Deklin. They are two great teammates who know how to make this project enjoyable. They also knew how to problem solve whenever we came across an obstacle. Our biggest obstacle was probably the amount of time we had to complete the project. not only are classes shorter, but we only had 2 days to build, one and a half days to test, and one more day to complete the packet. Our 2nd obstacle that make number difficult was that our fuel cells never worked. We would sped most of our time charging cells that would end up not even working. That took up 90% of our time. Then we also couldn't figure out how to set up the parallel circuits and how to test them. On the bright side, whenever we were working on the car, i was having a fun time, it wasn't total misery. Plus it was actually satisfying when i saw the car moved when we hooked it up to batteries. At least we got one thing to work. Now i just want to see who will end up winning the race(if we ever have one).
Conclusion
1. Using the measurements you made, compare and relate the four options you explored. Was the car best powered by a single fuel cell, a single solar module, two AAA batteries in series, or two AAA batteries in parallel?
Out of the 3 options our group was able to complete. we first found out that when the car was hooked up only to the solar panels, it didnt produce any voltage, current, power, etc. The car was completely dead. Now when we used the two AAA batteries, we found out that the ones in the series circuit produced more voltage, current, and power. The result for the ones in the series circuit were that it produced 5.6 volts, 140 ma, and .784 w. On the other hand the ones in the parallel circuit produced .18 volts, 25 ma, and .0045 w.
2. Did voltage, current, or power best describe the suitability of a power source?
I would have to say voltage. First off it was the easiest to measure. Also the power source automatically told you the voltage. another thing is that you can measure voltage anywhere on the circuit. And if there is no voltage, you can tell if the power source is suitable or not.
3. If you had many solar modules, how many of them would be needed to get the same performance from the car as the performance observed with two AAA batteries? Describe or sketch how would you connect the solar modules in terms of parallel and series circuits.
4. Describe and defend a system that you believe would best utilize a solar hydrogen system to meet the needs for an average driver.
The best system for an average driver would be to make all roads out of bulletproof solar panels with huge hydrogen centers every once a while along the road. You would also add heat lamps instead of regular lamps. therefore at night the road can still produce solar power with the heat of the lamp instead of the sun. you would need to make the solar panels bulletproof just in case something falls and breaks the road, or if there is just a car crash. finally you would have huge hydrogen centers so that they could act like present day gas station, where people can rest a little and let their cars energize.
5. How does a photovoltaic cell work? Record the source of your information.
a solar panel works by allowing photons, or particles of light, to knock electrons free from atoms, generating a flow of electricity. Solar panels actually comprise many, smaller units called photovoltaic cells. (photovoltaic simply means they convert sunlight into electricity.)
www.livescience.com/41995-how-do-solar-panels-work.html
6. Detail how electrolysis separates hydrogen and oxygen. How is electricity produced as the fuel cell allows the hydrogen to reunite in a bond with oxygen? Record the source of your information.
1) When electricity is introduced to water through two electrodes, a cathode (negative) and an anode (positive), these ions are attracted to the opposite charged electrode. Therefore the positively charged hydrogen ions will collect on the cathode and the negatively charged oxygen will collect on the anode.
2) When these ions come into contact with their respective electrodes they either gain or lose electrons depending on there ionic charge. so these ions balance their charges. this system isn't very efficient is because some of the electrical energy is converted into heat during the process. it is only 50%-70% efficient becuase most of the energy is lost due to the by product of heat.
http://www.instructables.com/id/Separate-Hydrogen-and-Oxygen-from-Water-Through-El/
1. Using the measurements you made, compare and relate the four options you explored. Was the car best powered by a single fuel cell, a single solar module, two AAA batteries in series, or two AAA batteries in parallel?
Out of the 3 options our group was able to complete. we first found out that when the car was hooked up only to the solar panels, it didnt produce any voltage, current, power, etc. The car was completely dead. Now when we used the two AAA batteries, we found out that the ones in the series circuit produced more voltage, current, and power. The result for the ones in the series circuit were that it produced 5.6 volts, 140 ma, and .784 w. On the other hand the ones in the parallel circuit produced .18 volts, 25 ma, and .0045 w.
2. Did voltage, current, or power best describe the suitability of a power source?
I would have to say voltage. First off it was the easiest to measure. Also the power source automatically told you the voltage. another thing is that you can measure voltage anywhere on the circuit. And if there is no voltage, you can tell if the power source is suitable or not.
3. If you had many solar modules, how many of them would be needed to get the same performance from the car as the performance observed with two AAA batteries? Describe or sketch how would you connect the solar modules in terms of parallel and series circuits.
4. Describe and defend a system that you believe would best utilize a solar hydrogen system to meet the needs for an average driver.
The best system for an average driver would be to make all roads out of bulletproof solar panels with huge hydrogen centers every once a while along the road. You would also add heat lamps instead of regular lamps. therefore at night the road can still produce solar power with the heat of the lamp instead of the sun. you would need to make the solar panels bulletproof just in case something falls and breaks the road, or if there is just a car crash. finally you would have huge hydrogen centers so that they could act like present day gas station, where people can rest a little and let their cars energize.
5. How does a photovoltaic cell work? Record the source of your information.
a solar panel works by allowing photons, or particles of light, to knock electrons free from atoms, generating a flow of electricity. Solar panels actually comprise many, smaller units called photovoltaic cells. (photovoltaic simply means they convert sunlight into electricity.)
www.livescience.com/41995-how-do-solar-panels-work.html
6. Detail how electrolysis separates hydrogen and oxygen. How is electricity produced as the fuel cell allows the hydrogen to reunite in a bond with oxygen? Record the source of your information.
1) When electricity is introduced to water through two electrodes, a cathode (negative) and an anode (positive), these ions are attracted to the opposite charged electrode. Therefore the positively charged hydrogen ions will collect on the cathode and the negatively charged oxygen will collect on the anode.
2) When these ions come into contact with their respective electrodes they either gain or lose electrons depending on there ionic charge. so these ions balance their charges. this system isn't very efficient is because some of the electrical energy is converted into heat during the process. it is only 50%-70% efficient becuase most of the energy is lost due to the by product of heat.
http://www.instructables.com/id/Separate-Hydrogen-and-Oxygen-from-Water-Through-El/